AI-based precision medicine mining of UK Biobank finds 13 human COVID risk genes and identifies 59 drugs with potential to improve survival in severe COVID-19 patients and sepsis
May 6, 2020
- 70 sepsis risk genes identified from UK Biobank, 61% of which are present specifically in severe COVID-19 patients (‘COVID risk genes’)
- 13 ‘COVID risk genes’ identified as known druggable targets
- 59 compounds and drugs identified with potential for accelerated drug discovery (‘repurposing’)
- Study also offers potential for identifying COVID-19 high risk biomarkers
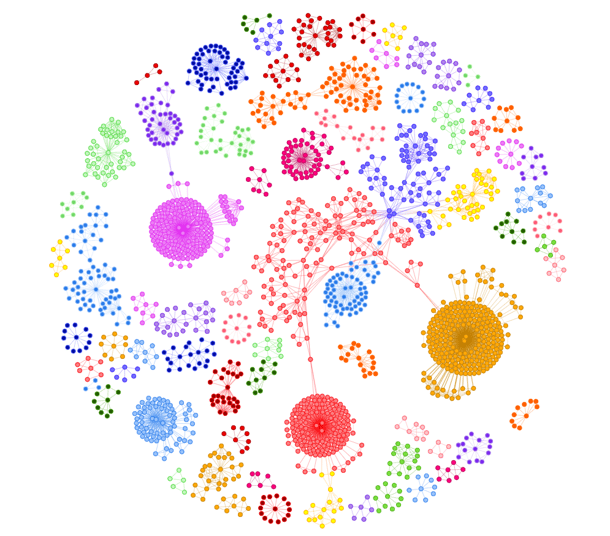 Caption: Disease architecture of the sepsis cohort generated by the precisionlife platform. Each circle represents a disease associated SNP genotype, edges represent co-association in patients, and colors represent distinct patient sub-populations or ‘communities’. To download this image, click here.
Caption: Disease architecture of the sepsis cohort generated by the precisionlife platform. Each circle represents a disease associated SNP genotype, edges represent co-association in patients, and colors represent distinct patient sub-populations or ‘communities’. To download this image, click here.
Data scientists from UK-headquartered AI precision medicine company, PrecisionLife, have used the Company’s proprietary AI enabled precision medicine platform to identify 59 repurposing drug candidates that could be used to develop new therapeutic strategies to increase the survival rate of patients who develop sepsis while suffering from severe COVID-19.
The new study, released today on Biorxiv sought to identify genetic risk factors for sepsis especially in the context of COVID-19, and to use these insights to identify existing drugs that might be used to treat life-threatening late-stage disease.
“Ours is the first study looking at host genomics and opportunities to treat later stage severe disease where host immune processes take over” said Dr Steve Gardner CEO of PrecisionLife.
Like the initial genomic studies on COVID-19 patients, previous analyses of sepsis patients have failed to identify more than a handful of genetic variants that predispose individuals to developing the disease. By providing deeper insights, this study identifies novel approaches and hope for new therapies.
PrecisionLife analyzed patient datasets compiled by UK Biobank to identify genes associated with sepsis, which are also found in severe COVID-19 patients. Sepsis is observed in 60% of severe COVID-19 patients and is a life-threatening condition with a mortality rate of approximately 20%.
The team identified mutations in 70 sepsis risk genes, 61% of which were also present specifically in severe COVID-19 patients. Several of the disease associated genetic signatures found in both sepsis and severe COVID-19 patients have previously been linked to cancer, immune response, endothelial and vascular inflammation and neuronal signalling.
13 of the sepsis risk genes, which the study shows are also COVID risk genes, are known to be druggable i.e. targeted by active chemical compounds used to treat these other diseases and therefore represent potential drug repurposing opportunities. The study went on to identify 59 compounds and drugs that are known to be active against these 13 targets. These could form the basis for future drug trials and repurposing projects. They could also offer potential as COVID-19 high risk biomarkers.
“Our high-resolution genomic analysis tools have allowed us to develop new insights into two serious and complex diseases for which new therapeutic options are urgently required. We hope that these will lead to better understanding of what drives sepsis in COVID-19 patients and result in new ways to treat seriously ill patients” said Dr Gardner.
PrecisionLife is disclosing its new insights and will be working with international collaborators to investigate therapeutic strategies that may help to reduce the high mortality rates in patients who develop sepsis with or without the context of COVID-19.
As more COVID-19 patient data become available in UK Biobank and other patient data sources, PrecisionLife will be able to analyze the clinical impact of these disease signatures in a larger group of patients.
For more information, please see www.precisionlife.com, or email covid-19@precisionlife.com.
Follow us on Twitter @precisionlifeAI and on LinkedIn www.linkedin.com/company/precisionlifeai
About PrecisionLife
PrecisionLife Ltd started in 2015, built on a shared vision to bring a new level of analytical capability to computational biology, genomic medicine and healthcare. Its powerful data analytics platform is built on a unique mathematical framework and over 30 years’ experience in delivering new technologies and products to enable the discovery of richer and more useful links between patients, disease, targets and drugs.
Headquartered in the UK, PrecisionLife also has operations in the US, Denmark and Poland.





