The clinical results of SCO-267 was presented at the Virtual 81st Scientific Sessions - American Diabetes Association: First report on the clinical data of a GPR40 full agonist in the phase 1 trial
June 28, 2021
SCOHIA presented the results of a phase 1 clinical trial for SCO-267, an orally bioavailable GPR40 full agonist, at the Virtual 81st Scientific Sessions - American Diabetes Association, June 25-29, 2021.
Presentation title
First in human, single and repeated-dose study of SCO-267, a GPR40 full agonist, in healthy adults and subjects with glucose intolerance
The presentation is available on the company's website (https://www.scohia.com/eng/).
Free fatty acid receptor 1 (GPR40) is a G protein-coupled receptor expressed in pancreatic islet and enteroendocrine cells, and SCO-267 is potentially a first in class full agonist of this receptor¹ˉ⁴. To the best of our knowledge, we are the first to reveal the effects of single- and multiple-oral doses of GPR40 full agonist in healthy adults and diabetic patients. SCO-267 was safe and well tolerated at all tested doses, and exhibited good potential for once daily dosing. In this study, we demonstrated that SCO-267-mediated full agonism of GPR40 stimulates the secretion of both islet and gut hormones, including insulin, glucagon, glucagon-like peptide 1, glucose-dependent insulinotropic polypeptide, and peptide YY in humans⁵. Oral administration of SCO-267 remarkably decreased fasting hyperglycemia and improved glycemic control during an oral glucose tolerance test in diabetic patients, without inducing hypoglycemia. These results collectively suggest a clinical potential of SCO-267 in treating diabetes. Based on its feature of stimulating islet and gut hormones, which regulate metabolism and body weight, SCO-267 may exhibit clinical benefits in treating diabetes, obesity, and nonalcoholic steatohepatitis. Therefore, SCO-267 is currently being prepared for a phase 2 clinical trial.
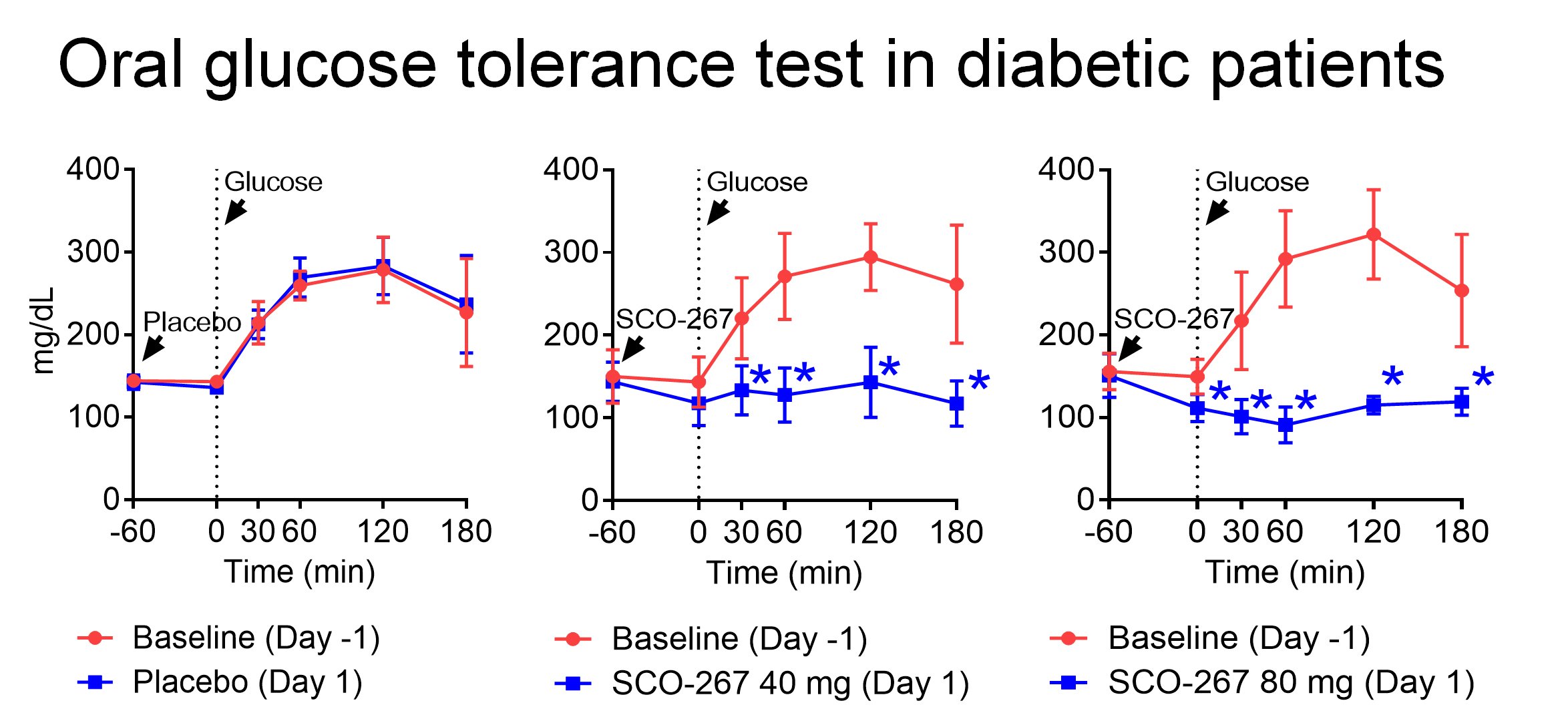 Figure. Effects of a single oral dose of SCO-267 on glycemic control during the oral glucose tolerance test in diabetic patients.
Figure. Effects of a single oral dose of SCO-267 on glycemic control during the oral glucose tolerance test in diabetic patients.
(n=4, 6, and 6 for placebo, SCO-267 40 mg, and 80 mg, respectively). *95% confidence interval did not cross zero at the indicated time points; SCO-267 versus placebo using the baseline (day-1)-adjusted value. Mean±SD.
References
1. Koyama, R., Ookawara, M., Watanabe, M. & Moritoh, Y. Chronic Exposure to SCO-267, an Allosteric GPR40 Full Agonist, Is Effective in Improving Glycemic Control in Rats. Mol Pharmacol 99, 286-293, doi:10.1124/molpharm.120.000168 (2021).
2. Furukawa, H. et al. Design and Identification of a GPR40 Full Agonist (SCO-267) Possessing a 2-Carbamoylphenyl Piperidine Moiety. J Med Chem 63, 10352-10379, doi:10.1021/acs.jmedchem.0c00843 (2020).
3. Ookawara, M., Matsuda, K., Watanabe, M. & Moritoh, Y. The GPR40 Full Agonist SCO-267 Improves Liver Parameters in a Mouse Model of Nonalcoholic Fatty Liver Disease without Affecting Glucose or Body Weight. J Pharmacol Exp Ther 375, 21-27, doi:10.1124/jpet.120.000046 (2020).
4. Ueno, H. et al. SCO-267, a GPR40 Full Agonist, Improves Glycemic and Body Weight Control in Rat Models of Diabetes and Obesity. J Pharmacol Exp Ther 370, 172-181, doi:10.1124/jpet.118.255885 (2019).
5. Insulin and glucagon are secreted by pancreatic islet, and glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic peptide (GIP), and peptide YY (PYY) by enteroendocrine cells, to regulate metabolism and body weight.
About SCOHIA PHARMA, Inc.:
SCOHIA PHARMA, Inc. is a drug discovery bioventure focusing on the field of lifestyle-related diseases such as cardiovascular, metabolic, and renal diseases. Our R&D team has a rich pipeline and track record in each stage of drug development, including compound discovery, drug evaluation, and clinical development, which makes us special. For detailed information about SCOHIA PHARMA, Inc., please visit https://www.scohia.com/eng/.





